Cell processing and functional testing
Cell processing and functional testing
Stem Cells Isolation
Stem cell isolation is a fundamental step in regenerative medicine and cell-based therapies, as it provides researchers with the raw biological material needed to study developmental biology, model diseases, and design therapeutic strategies. Stem cells can be derived from various sources, including bone marrow, adipose tissue, peripheral blood, umbilical cord blood, and embryonic or induced pluripotent stem cells (iPSCs). The choice of source depends on accessibility, ethical considerations, and intended applications. Isolation techniques aim to separate stem cells from a heterogeneous population of cells while preserving their viability, multipotency, and proliferative capacity.
Several strategies have been developed to achieve efficient stem cell isolation. Density gradient centrifugation is commonly used for separating mononuclear cells, while immunomagnetic bead sorting and fluorescence-activated cell sorting (FACS) allow for highly specific selection based on surface markers such as CD34 for hematopoietic stem cells or CD105/CD73/CD90 for mesenchymal stem cells. Advances in microfluidics and label-free methods, such as size-based filtration or dielectrophoresis, are providing new opportunities for gentle and scalable isolation without the need for extensive labeling. Each method has trade-offs between yield, purity, speed, and cost, and the selection of technique often depends on the downstream application—whether for research, preclinical studies, or clinical therapy.
Despite significant progress, stem cell isolation faces important challenges. One major limitation is heterogeneity—stem cell populations are rarely homogeneous, and isolating pure subsets while maintaining functionality is difficult. Marker-based selection can be problematic since no single marker is universally specific for stem cell populations, leading to contamination with other cell types. The process itself can induce stress or alter cell behavior, reducing viability or differentiation potential. Scaling up isolation methods for clinical-grade applications presents further difficulties, as techniques that work in the laboratory may not be suitable for large-scale or GMP-compliant production. Ethical and regulatory considerations, particularly for embryonic stem cells, also continue to shape how and where certain isolation strategies can be applied.
The workflow of stem cell isolation usually begins with tissue collection, followed by mechanical or enzymatic dissociation to release cells. The resulting suspension is then subjected to enrichment and purification steps such as density gradients, magnetic sorting, or flow cytometry. Isolated cells are subsequently characterized for viability, phenotype, and stemness markers before being expanded in culture or used directly in experiments. In clinical contexts, additional steps such as sterility testing, cryopreservation, and quality control are integrated to ensure safety and reproducibility.
By improving efficiency, reducing stress on cells, and developing scalable protocols, stem cell isolation continues to evolve as a cornerstone of regenerative medicine. The refinement of isolation methods not only enhances research reproducibility but also paves the way for safe and effective therapeutic applications, bringing stem cell–based treatments closer to routine clinical practice.
Flow Cytometry
Flow cytometry is one of the most powerful and versatile tools in modern biomedical research, offering the ability to analyze millions of individual cells in suspension within minutes. Its importance lies in providing high-throughput, multiparametric data that cannot be achieved with conventional techniques such as microscopy. By combining speed, precision, and statistical robustness, flow cytometry has become essential not only in basic science but also in clinical diagnostics and biopharmaceutical development. For example, immunophenotyping of T-cells, B-cells, and NK cells has transformed immunology research, while the routine monitoring of CD4+ T-cells in HIV patients demonstrates its clinical impact. The technology is equally valuable for cell cycle analysis, apoptosis detection, stem cell characterization, and high-throughput drug screening, making it a true workhorse across both academic and industrial laboratories.
Despite its tremendous potential, flow cytometry comes with notable challenges. Instruments are expensive and require highly trained operators to design experiments, optimize staining panels, and interpret complex datasets. Multiparametric analysis often introduces issues such as spectral overlap between fluorophores, which necessitates careful compensation and calibration. Sample preparation is another critical step, as improper handling may lead to cell clumping, artifacts, or even loss of sensitive populations. Moreover, variability between instruments and laboratories can complicate data reproducibility, making standardization a key concern for regulatory acceptance and translational applications.
The process of flow cytometry follows a series of well-established steps that together ensure reliable results. Cells are first harvested and suspended in a suitable buffer, followed by staining with fluorochrome-conjugated antibodies or fluorescent dyes that target specific surface or intracellular markers. Once prepared, the suspension is introduced into the flow cytometer, where each cell passes individually through a focused laser beam. Light scattering provides information on cell size and internal complexity, while emitted fluorescence signals are collected to reveal marker expression. With appropriate software, researchers apply gating strategies to identify distinct subpopulations, quantify cell frequencies, and extract meaningful biological insights. To ensure data quality, instruments are routinely calibrated with fluorescent beads and strict quality-control protocols.
By addressing the challenges of cost, complexity, and standardization, laboratories and companies can fully harness the potential of flow cytometry. Its ability to simultaneously capture structural, functional, and molecular features at the single-cell level continues to drive breakthroughs in cancer immunology, stem cell research, and therapeutic development. As innovations such as spectral cytometry and automated analysis expand its capabilities, flow cytometry remains a cornerstone technology at the intersection of research, clinical practice, and industrial biomedicine.
Stem Cell Differentiation Assay Methods
Stem cell differentiation assays are essential techniques used to evaluate the potential of stem cells to develop into specific cell types, such as osteocytes, adipocytes, or neurons. These assays are crucial in regenerative medicine, drug discovery, and tissue engineering because they confirm stem cell potency and guide experimental and therapeutic applications.
The main advantages of these assays include providing functional insights into stem cell biology, validating stem cell lines, and optimizing culture conditions for reproducible results. They are widely applied in research, preclinical studies, and industrial stem cell-based products.
In practice, laboratories often face challenges such as inconsistent differentiation efficiency, cell contamination, and batch-to-batch variability. Improper media composition, suboptimal induction protocols, or incorrect timing can lead to incomplete differentiation or heterogeneous populations. These issues are addressed by careful selection of differentiation factors, strict aseptic techniques, and standardized induction protocols. Our consulting and training services guide teams in designing reliable differentiation assays, troubleshooting variability, and ensuring reproducible results.
Stem cell differentiation assays remain indispensable for verifying stem cell functionality. For hands-on training or specialized consulting in stem cell differentiation, contact our expert team to optimize your workflows.
Cell Culture
Stem cell differentiation assays are essential techniques used to evaluate the potential of stem cells to develop into specific cell types, such as osteocytes, adipocytes, or neurons. These assays are crucial in regenerative medicine, drug discovery, and tissue engineering because they confirm stem cell potency and guide experimental and therapeutic applications.
The main advantages of these assays include providing functional insights into stem cell biology, validating stem cell lines, and optimizing culture conditions for reproducible results. They are widely applied in research, preclinical studies, and industrial stem cell-based products.
In practice, laboratories often face challenges such as inconsistent differentiation efficiency, cell contamination, and batch-to-batch variability. Improper media composition, suboptimal induction protocols, or incorrect timing can lead to incomplete differentiation or heterogeneous populations. These issues are addressed by careful selection of differentiation factors, strict aseptic techniques, and standardized induction protocols. Our consulting and training services guide teams in designing reliable differentiation assays, troubleshooting variability, and ensuring reproducible results.
Stem cell differentiation assays remain indispensable for verifying stem cell functionality. For hands-on training or specialized consulting in stem cell differentiation, contact our expert team to optimize your workflows.
Cell proliferation
Cell culture and expansion are foundational techniques for generating sufficient numbers of healthy cells for research, therapy, or industrial applications. Maintaining optimal growth conditions is critical for cell viability, functionality, and consistency in downstream applications.
The advantages of well-executed cell culture include controlled growth, scalability, and the ability to manipulate cell conditions for experimental or therapeutic purposes. Applications range from basic research and drug screening to cell therapy and regenerative medicine.
Common real-world challenges include contamination (bacterial, fungal, or mycoplasma), cell senescence, and variable growth rates. Using suboptimal media, improper incubator conditions, or inconsistent handling can compromise cell health. Solutions include implementing strict aseptic protocols, using optimized media formulations, monitoring cell morphology, and training staff in best practices. Our consulting and training services help labs improve culture reliability, scale-up efficiency, and quality control processes.
Cell culture and expansion are the backbone of all cellular research and therapy. For expert consulting or hands-on workshops in cell culture, connect with our team to ensure robust and reproducible outcomes.
Cell Counting
Cell counting is a fundamental technique used to determine cell concentration and viability, providing critical information for experimental planning, seeding cultures, and quality control. Accurate cell counts are essential for reproducible results in research and production.
The advantages include precise dosing, monitoring proliferation, and optimizing resource use. This technique is universally applied in drug screening, stem cell studies, and biomanufacturing.
Laboratories often face challenges such as clumped or dead cells leading to inaccurate counts, manual counting errors, and instrument calibration issues. These problems can be solved through proper sample preparation, using automated counters or viability dyes, and regular equipment maintenance. Our consulting and training services help labs implement reliable counting protocols, minimize human error, and standardize quality control procedures.
Accurate cell counting is essential for reproducible research and clinical-grade cell production. For training or consulting on advanced cell counting techniques, our team provides practical guidance to optimize accuracy and efficiency.
Cell Freeze (Backup Preparation) / Defreeze
Cell freezing and thawing are critical procedures for long-term cell storage, quality assurance, and experimental reproducibility. Proper cryopreservation ensures cell viability, phenotype maintenance, and readiness for downstream applications.
The advantages include creating reliable backup stocks, maintaining genetic integrity, and enabling flexible research planning. Applications range from stem cell banking, biobanking, and regenerative medicine to routine laboratory experiments.
Common challenges include reduced viability after thawing, ice crystal formation damaging cells, and variability in recovery between batches. These issues can be addressed by using optimized cryoprotectants, controlled-rate freezing, and standardized thawing protocols. Our team provides consulting on cryopreservation strategies, hands-on training in freeze/thaw techniques, and troubleshooting to maximize post-thaw recovery and functionality.
Effective cell freezing and thawing is essential for consistent and reliable cell-based research. For expert guidance or hands-on workshops in cell cryopreservation, reach out to our experienced team today.
Cytotoxicity Assay
MTT Assay
The MTT assay is a widely used method to assess cell viability and metabolic activity. It relies on the reduction of the yellow MTT reagent into insoluble purple formazan crystals by mitochondrial enzymes in living cells. This assay is critical in drug development, cytotoxicity evaluation, and stem cell research, as it provides a quantitative measure of cell health.
One of the main advantages of the MTT assay is its simplicity, cost-effectiveness, and reproducibility, making it suitable for high-throughput screening. It is widely applied in pharmaceutical testing, biomaterial evaluation, and cancer research to assess the effects of compounds on cell proliferation.
In practice, labs often encounter challenges such as incomplete solubilization of formazan crystals, compound interference due to inherent color, and well-to-well variability. These issues can lead to inaccurate readings and misinterpretation of results. Solutions include optimizing solubilization protocols, using proper controls, standardizing cell seeding densities, and training staff on precise handling. Our team provides consulting and hands-on training to overcome these challenges, ensuring reliable and reproducible outcomes.
The MTT assay remains a cornerstone technique for cytotoxicity evaluation. For expert guidance, training, or protocol optimization in MTT assays, contact our experienced team today.
Alamar Blue Assay
The Alamar Blue assay is a sensitive, non-toxic method for assessing cell viability and metabolic activity. It uses resazurin, a blue dye, which is reduced to resorufin, a fluorescent compound, by viable cells. This allows real-time monitoring of cell proliferation and cytotoxicity without harming the cells.
Its main advantages include compatibility with high-throughput formats, non-destructiveness (enabling repeated measurements on the same sample), and high sensitivity. Alamar Blue is extensively used in drug discovery, nanomaterial evaluation, stem cell studies, and toxicity screening.
Despite its utility, laboratories may face challenges such as variable incubation times affecting signal intensity, reagent instability, and interference from colored media or test compounds. These issues can compromise accuracy if not properly addressed. Solutions include standardizing assay timing, using freshly prepared reagents, applying appropriate controls, and optimizing sample handling. Our consulting and training services help laboratories implement robust Alamar Blue protocols, troubleshoot variability, and interpret results with confidence.
The Alamar Blue assay provides a flexible, reliable approach to measuring cell viability. For hands-on training or consulting in Alamar Blue assays, reach out to our expert team to optimize your workflows.
Migration& invasive Assay
Invasive Assay
An invasion assay is a laboratory technique used to measure the ability of cells to invade through a barrier that mimics the extracellular matrix (ECM) and basement membranes in the body.
The purpose of the invasion assay is related to investigating metastasis, inflammation, and embryonic development, cancer research, drug discovery, and basic biology.
Boyden Chamber Assay (Transwell Invasion Assay) is the gold standard for this test, which uses a special insert with a porous membrane. Matrigel is an important component in this assay, which consists of a gelatinous protein mixture secreted by mouse tumor cells. Before the experiment, a thin layer of Matrigel is applied to the top of the membrane in the insert, creating a barrier that cells must actively degrade and invade through.
The procedure of the invasion assay includes placing a well containing cell culture medium that often contains a chemoattractant—a substance that lures cells to move toward it (e.g., a growth factor like FBS). In the upper chamber, cells being tested are seeded in a serum-free medium and after 24-48 hours of incubation, the chemoattractant in the lower chamber creates a concentration gradient. The cells are motivated to move downward toward the attractant. In conclusion, non-invasive cells remain on the top surface of the matrigel and invasive cells are found on the bottom surface of the membrane. By staining the cells and counting the number that have successfully invaded through to the bottom side, are done.
The entire process is fraught with potential challenges that can lead to high variability, no invasion, or false results. Some of the problems are related to Matrigel handling and coating because it gels at room temperature (22-35°C) but is liquid at 4°C. Premature gelling during handling can create an uneven, lumpy coating that is either impenetrable or inconsistently thick. For solving this problem, keeping all procedures in a cold condition and optimizing the concentration is highly recommended. The Wiping Step is the most hands-on and variable. Incomplete wiping leaves non-invasive cells behind, inflating the invasion count. Over-wiping can damage the membrane and dislodge the cells you want to count. To fix this problem, using a cotton-tipped swab moistened with PBS and checking the wiping under a microscope to ensure all non-invasive cells are removed is very important.
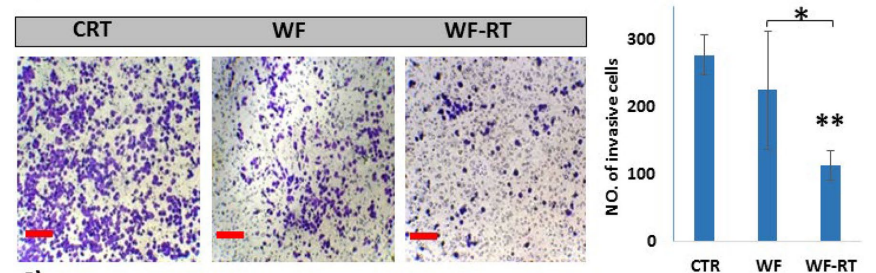
Effects of WFs on invasion BC cell line (MDA-MB 231) and in human-derived tumor spheroids. The images of the cells in the upper chamber of the Transwell assay and the graph present the percentage of cell migration. https://doi.org/10.1038/s41598-022-11023-z
The invasion assay is a cornerstone of cancer metastasis research. While its technical challenges are significant, primarily centered on the variability of Matrigel, cell health, and the manual wiping/counting steps, they are not insurmountable. By understanding these potential pitfalls and implementing the problem-solving strategies, you can generate reliable and meaningful data that accurately reflects the invasive potential of your cells. Our consulting team specializes in troubleshooting these types of problems exactly.
Scratch assay
The “Scratch” or “wound” test is a basic concept in which a monolayer of cells is observed and measured over time as the cells move to close the gap. It’s one of the oldest and most straightforward methods to study this critical biological process. The importance of the scratch assay is to investigate cell migration in many physiological and pathological conditions, such as wound healing, embryonic development, immune response, and cancer metastasis. The scratch assay is a widely used, simple, and cost-effective technique to study collective two-dimensional cell migration in vitro, as it requires minimal equipment and allows for the straightforward visualization and quantification of how fast cells close the wound, providing direct insight into processes like wound healing, metastasis, and the effects of drugs or genes on cell motility. However, its major limitation is that it only models a specific type of mechanical wounding and fails to recapitulate the complex three-dimensional microenvironment and cell-matrix interactions present in actual physiological or pathological processes.
The procedure of the scratch assay includes cell culture with a confluent (100%) monolayer that covers the entire surface. Creating the “Scratch” as the key step, in which the monolayer is scratched in a straight line to create a cell-free gap. The dish is gently washed with a buffer solution to remove the dislodged cells and debris. In this step, the treatment is added to see its effect on migration. Eventually, imaging, monitoring, and analysis are done by placing them under a microscope and taking pictures at regular intervals (e.g., every 12, 24, and 48 hours). The images are analyzed using software (like ImageJ) to measure the width of the scratch at each time point.
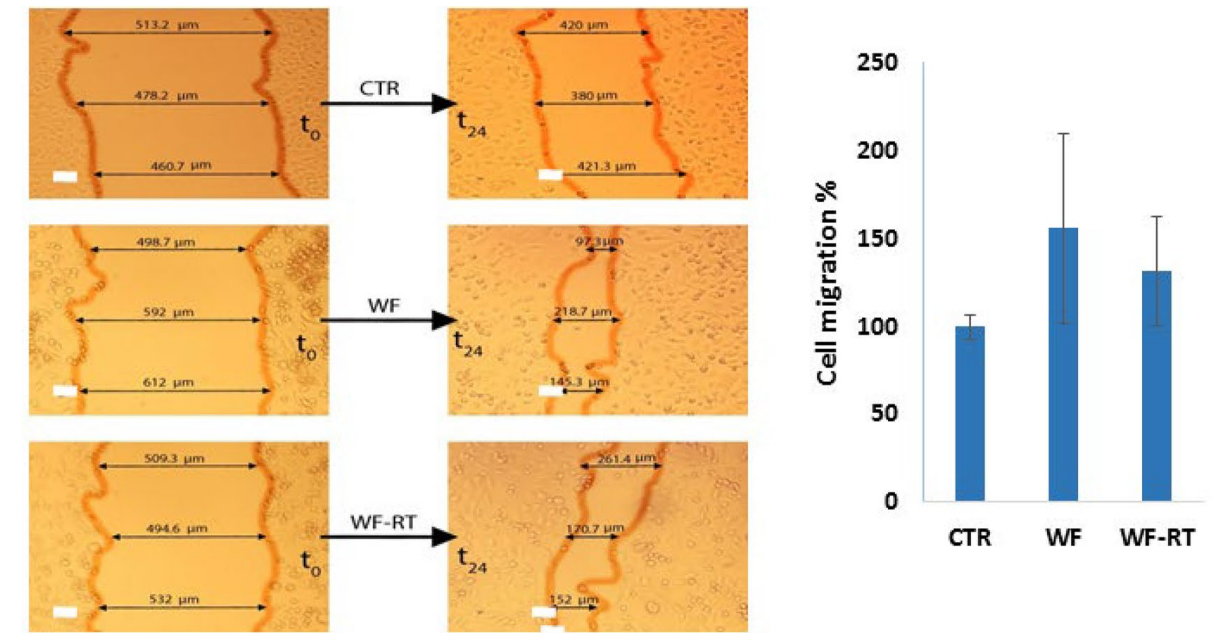
Effects of WFs on migration of BC cell line (MDA-MB 231) and in human-derived tumor spheroids. Images and graphs related to scratch assay (wound healing compared to negative control in 0 and 24 h). The graph presents the percentage of migrated cells. https://doi.org/10.1038/s41598-022-11023-z
The challenge in scratch assay can be broken down into three phases: scratch creation, post-scratch handling, and imaging/analysis.
The most important issue in scratch creation is related to inconsistent scratch width, which makes quantification unreliable. To solve this problem, practicing and repeating the technique and using a skilled guide can be useful.
The most important issue in post-scratch handling is related to cell death or detachment and proliferation vs. migration. Therefore, to decrease cell death, all washing and media changes should be done gently. To investigate migration, Mitomycin-C is used to inhibit or halt cell division without affecting migration.
The most important issue in imaging and analysis is related to subjectivity in analysis. Therefore, to remove this issue, using automated software, such as ImageJ (Fiji) and blind analysis, is highly recommended.
The scratch assay is a classic example of a technique where the art of the researcher is as important as the science. Careful attention to these challenges will lead to robust and publishable data. Hence, our consulting and training programs for scratch assay are designed to help researchers overcome these exact bottlenecks.
Tube Formation Assay
The Tube Formation Assay is used to study angiogenesis and the process of forming new blood vessels from pre-existing ones. By seeding endothelial cells on top of the Matrigel as a basement membrane extract (BME), investigating cells attach, migrate, align, and form an interconnected is explained. The resulting structures are then quantified by measuring metrics like total tube length, number of branch points, number of meshes, and total mesh area.
Advantages of the Tube formation assay include rapid and high-throughput capable, highly reproducible (When Optimized), excellent for phenotypic screening, relatively simple and inexpensive, amenable to quantification, and reduces animal Use (3Rs Principle). However, a significant limitation of this assay is its oversimplification of the complex in vivo angiogenic microenvironment, which can lead to results that may not fully predict in vivo efficacy or biological complexity.
The application of tube formation assay is confirmed to assess the pro- or anti-angiogenic effects of various compounds, drugs, gene modifications, or conditioned media from other cell types. This procedure is also used for screening potential cancer therapeutics, studying vascular biology in diseases like diabetes, and testing the efficacy of regenerative medicine approaches designed to promote vascularization in engineered tissues.
The tube formation assay procedure begins by thawing and gently pipetting a basement membrane matrix, like Matrigel, into the wells of a pre-chilled multi-well plate, which is then incubated to allow the matrix to polymerize into a solid gel. Subsequently, endothelial cells, such as Human Umbilical Vein Endothelial Cells (HUVECs), are seeded as a single-cell suspension on top of the set gel and incubated. Over a period of several hours (typically 4-18 hours), the endothelial cells will spontaneously form interconnected, mesh-like structures that mimic early capillary networks, which are finally visualized and quantified using an inverted light microscope and image analysis software to measure parameters like total tube length, number of junctions, and mesh area.
One of the important biological challenges in interpretation is that the assay doesn’t tell the whole story of angiogenesis. It measures differentiation, not proliferation. To solve this issue, complementing the tube assay with a proliferation assay (e.g., MTT, BrdU) is highly recommended.
By understanding, training, and addressing the challenges through our team, you can transform the tube formation assay from a finicky art into a robust and powerful quantitative tool for angiogenesis research.
Apoptosis Assay
Based on the central role of apoptosis, accurately detecting and quantifying apoptosis is vital in fields like cancer research, neurobiology, immunology, and drug discovery. Because it’s a multi-step process with distinct biochemical and morphological changes (cell shrinkage, membrane blebbing, DNA fragmentation, etc.), no single assay can capture every aspect. This is why the choice of assay and its interpretation are critical.
The main advantage is the ability to precisely identify and measure a specific, fundamental biological process. This breaks down into several key benefits of specificity over viability assays, mechanistic insight, high sensitivity and quantification, and versatility with multiple readouts.
There are some important applications of the apoptosis assay, including drug discovery, toxicology, and basic research.
Apoptosis assays are categorized into several groups, including assays based on Phosphatidylserine (PS) Externalization, which is one of the earliest and most reliable markers. Assays based on caspase activation that target the core apoptotic machinery and divided into immunoblotting, fluorometric, and caspase-specific antibodies. Assays based on DNA fragmentation, which is a classic hallmark of late apoptosis and consists of TUNEL assay (Terminal deoxynucleotidyl transferase dUTP Nick End Labeling) and DNA laddering. Assays based on mitochondrial changes that detect the early initiation phase of the intrinsic apoptotic pathway. Eventually, morphological assays, which are the most straightforward method used for initial confirmation, are detected by light microscopy and fluorescence microscopy with DNA-binding dyes such as Hoechst 33342 and DAPI.
The procedure of apoptosis assays is used to detect and quantify the characteristic biochemical and morphological changes that occur in programmed cell death.
The general procedure begins by inducing apoptosis in a cell population (e.g., with drugs or radiation) and preparing the cells for analysis. Depending on the specific assay chosen, the technique involves staining cells with fluorescent dyes or antibodies that target key apoptotic markers, such as the externalization of phosphatidylserine (detected by Annexin V binding), the disruption of the mitochondrial membrane potential, the activation of executioner caspases, or the fragmentation of nuclear DNA. After staining, the cells are analyzed using specialized instrumentation like flow cytometry for high-throughput, quantitative data or fluorescence microscopy for visual confirmation of cellular morphology.
The main challenge stems from the fact that apoptosis is not a single event but a dynamic multi-pathway process with distinct stages:
Early Apoptosis: Initiation phase. Key events include phosphatidylserine (PS) exposure on the outer leaflet of the cell membrane and mitochondrial membrane depolarization.
For detecting this step, Annexin V / PI is used, which is better used as a dual-color assay.
Mid Apoptosis: Execution phase. Activation of caspase enzymes.
For detecting this step, Caspase Activity is used with specific caspase substrates/inhibitors.
Late Apoptosis: Degradation phase. DNA fragmentation, membrane blebbing, and formation of apoptotic bodies. For detecting this step, TUNEL assay is used.
Secondary Necrosis: Final phase. Loss of membrane integrity, which can look like primary necrosis. Detecting membrane integrity cannot easily distinguish a late apoptotic cell. Hence, multi-parameter assays are the gold standard, such as Annexin V/PI dual staining and Propidium Iodide (PI) for solving this problem.
Our team offers hands-on training and consulting to troubleshoot and implement the solutions to apoptosis assays. By addressing these challenges, we help researchers and companies significantly increase the accuracy and reliability of their apoptosis data.
TUNEL Assay
The TUNEL assay (Terminal deoxynucleotidyl transferase dUTP Nick End Labeling) is a specialized technique for detecting DNA fragmentation in apoptotic cells. It is crucial for evaluating programmed cell death in response to drugs, radiation, or toxic compounds, and for studying apoptosis in developmental biology and cancer research.
The main advantages of TUNEL assays include high specificity for apoptosis detection, compatibility with tissue sections and cultured cells, and the ability to provide both qualitative and quantitative data. It is widely applied in toxicology, cancer research, and stem cell studies to confirm cytotoxic effects at the molecular level.
However, laboratories frequently encounter challenges such as false positives due to improper fixation or permeabilization, enzymatic reaction variability, and difficulty distinguishing apoptotic from necrotic cells. Addressing these issues requires careful optimization of fixation, permeabilization, and labeling steps, alongside rigorous controls. Our team provides consulting and hands-on training to optimize TUNEL protocols, minimize background signals, and ensure reproducible, reliable apoptosis detection.
TUNEL assays remain indispensable for studying apoptosis and cytotoxicity at the molecular level. For expert consulting or hands-on training in TUNEL assays, contact our team to enhance accuracy and reliability in your experiments.
Sign up to receive our latest updates
Get in touch
Call us directly?
Address:
Email: